Test Covid 19 Ag

Bioeasy 2019 ncov ag fluorescence test is a fast and reliable test for 2019 ncov.
Test covid 19 ag. It has been authorized by the fda under an emergency use authorization for use by authorized laboratories. For more product details. Bioeasy 2019 ncov ag fluorescence test is a fast and reliable test for 2019 ncov. Please understand that we cannot respond to enquiries from non professional users.
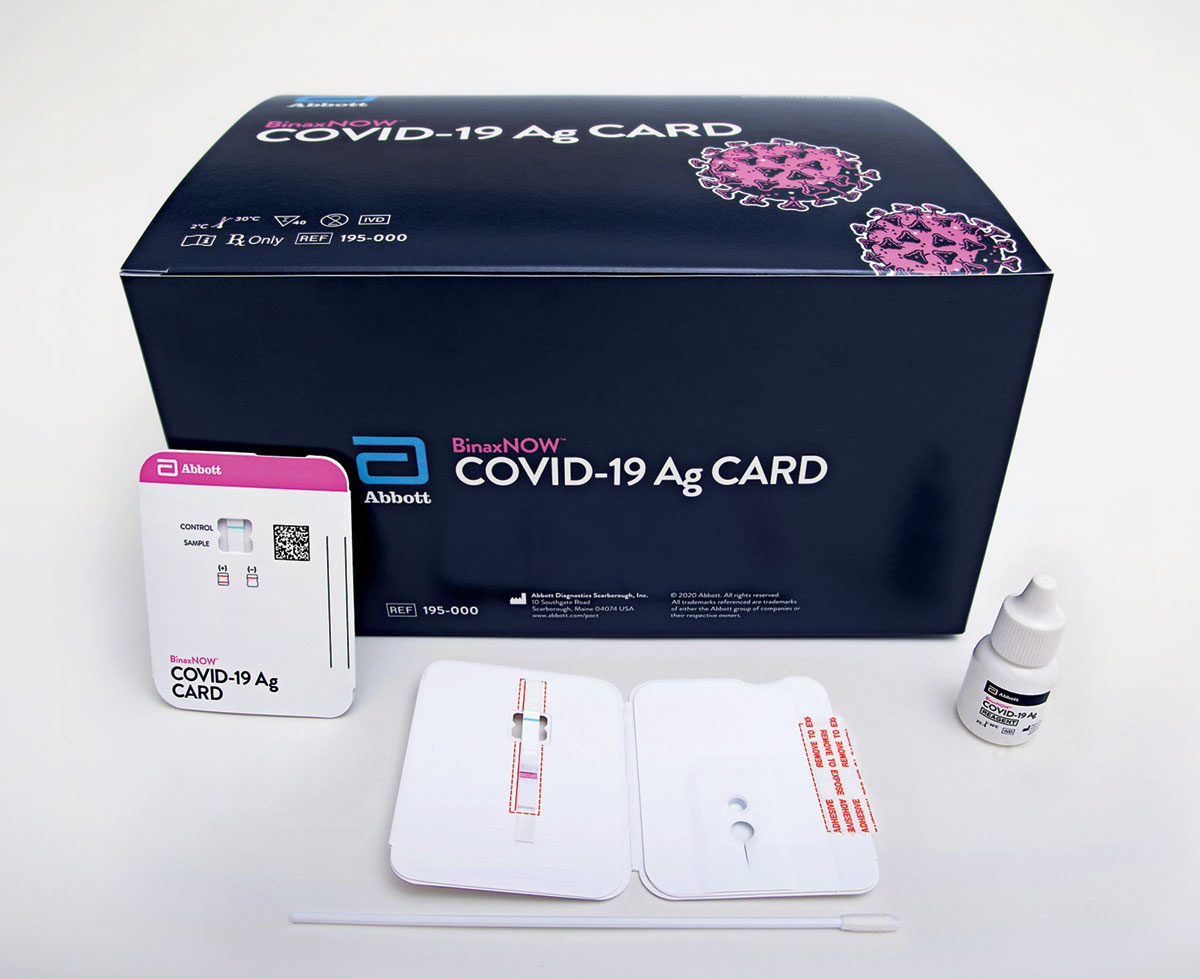
Called covid 19 ag respi strip the test is only commercially available to healthcare services. To deal with the spread of the coronavirus disease 2019 covid 19 coris bioconcept has worked hard these past few weeks to develop a rapid antigen test that can be used for the detection of sars cov 2 in nasopharyngeal secretions. Standard q covid 19 ag test is a rapid chromatographic immunoassay for the qualitative detection of specific antigens to sars cov 2 present in human nasopharynx. The binaxnow covid 19 ag card eua has not been fda cleared or approved.
Check the test device and the desiccant pack in the foil pouch. Increase your test capacities and stop the outbreak with an accurate antigen testing for severe and critical stages. Our nadal covid 19 igg igm and ag rapid tests are approved for professional users i e. You do not need a test unless recommended or required by your healthcare provider or public health official.
Fast results within 15 30 mins easy to use. If you do not have covid 19 symptoms and have not been in close contact with someone known to have sars cov 2 infection meaning being within 6 feet of an infected person for at least 15 minutes. It is not for sale to the general public. Covid 19 ag test tm 2 c 36 f 30 c 86 f test device individually in a foil pouch with desiccant extraction buff er tube nozzle cap sterile swab instructions for use carefully read instructions for using the standard q covid 19 ag test.
This interim guidance is intended for clinicians who order antigen tests receive antigen test results and or perform point of care testing as well as for laboratory professionals who perform antigen testing in a laboratory setting or at the point of care and. With the addition of deep sputum or nasal swab it can result in 10 minutes b. The test has been authorized only for the detection of proteins from sars cov 2 not for any other viruses or pathogens and is only authorized for the duration of the declaration that circumstances exist. Antigen tests can be used in a variety of testing strategies to respond to the coronavirus disease 2019 covid 19 pandemic.